Latest research has been published in Nature
Methane Oxidation to Ethanol by a Molecular Junction Photocatalyst

The 18th National Solar Photochemistry and Photocatalysis Conference
Prof. Tang was invited to deliver a Plenary Lecture

2025 Annual Meeting
Tang Group Wraps Up Annual Meeting with Focused Discussions
Introduction
The Laboratory of photon-phonon co-driven catalysis led by Professor Junwang Tang, has established a new concept of photon-phonon co-driven catalysis, which is employed to achieve efficient conversion of small molecules. In parallel, the group also focuses on plastic waste recycling by microwave catalysis and biomass transformation through multi-energy-coupled catalytic technologies.
The group's main research projects include: ammonia synthesis, hydrogen production, biomass conversion, plastic depolymerization, natural gas conversion and multi-scale ultrafast spectroscopy.
Research
Ammonia synthesis
1. To develop advanced thermal catalysts for efficient ammonia synthesis under mild conditions, aiming to overcome the energy-intensive limitations of the Haber-Bosch process through innovative catalyst design.
2. To pioneer photon-phonon co-driven catalytic systems that synergistically combine light and thermal energy for efficient nitrogen fixation. This enables ammonia synthesis from nitrogen and water (or hydrogen) at significantly lower temperatures and pressures than conventional methods, offering a sustainable pathway toward decentralized green ammonia production.

Solar-driven hydrogen production
To design highly efficient and stable photon-phonon co-driven catalytic systems to achieve solar-driven water splitting or methanol reforming for green hydrogen production, thereby contributing to the development of the hydrogen energy industry and the realization of carbon neutrality goals.

Photon-phonon co-driven catalysis for biomass conversion
To develop an efficient photon–phonon co-driven catalysis strategy for the selective and efficient conversion of biomass, including the conversion of glycerol into glyceraldehyde, glycolaldehyde, or lactic acid and methanol into value-added chemicals under mild conditions.
Microwave-assisted catalysis for waste recycling
To employ microwave-catalysis technology for the efficient degradation and conversion of waste plastics (e.g., PE, PP, PET), enabling the targeted production of high-value chemicals or fuels to promote white pollution control and carbon resource recycling.
Photon-phonon co-driven catalysis for natural gas conversion
To develop novel catalytic materials and reaction systems for efficient conversion of natural gas into high-value chemicals or clean fuels at low temperatures, offering a low carbon route for natural gas resource utilization.

Multi-scale in-situ spectroscopy
To develop the in-situ/ operando multifunctional time-resolved (fs-ms) spectroscopies, aiming to unveil the reaction pathways in thermal catalysis, electrocatalysis and photon-phonon co-driven catalysis.

Members
Professor

Professor Tang is a pioneer in employing multiple energy sources to activate small molecules (e.g., H2O, N2, CH4, CO2). He develops low carbon catalytic systems for the conversion of these molecules to high-value chemicals and employs microwave catalysis to depolymerize solid waste, enabling resource recycling. He is also dedicated to investigating the reaction mechanisms of photo/thermal/electro-catalysis using multi-modal time-resolved spectroscopies.
Professor Tang has authored more than 250 papers in prestigious international journals such as Nature, Nature Catalysis, Nature Energy, Nature Materials, and Nature Sustainability. His work has been cited over 36,000 times, earning him an h-index of 94 (Google Scholar). He has been granted 25 patents and has been recognized as a Clarivate Analytics Highly Cited Researcher on multiple occasions. He also serves as an Editor or Associate Editor for 6 international journals, including Applied Catalysis B: Environment and Energy, Chinese Journal of Catalysis, and Energy & Environmental Science: Solar.
Google Scholar link: https://scholar.google.com/citations?user=S0jdSPEAAAAJ&hl=zh-CN&oi=sra
Postdoctoral Researchers

Photon-phonon co-driven catalysis for ammonia synthesis

Photon-phonon co-driven methane conversion

Photon-phonon co-driven catalysis for methanol conversion

Thermal catalysis for ammonia decomposition

Mechanistic investigation of photothermal catalytic systems using multi-scale time-resolved spectroscopy

Photon-phonon co-driven oxidation of nitrogen to nitrite

Microwave catalysis for convertion of waste plastics into high-value products

H2O2 synthesis coupled with biomass conversion

Photocatalytic ammonia synthesis

Thermal catalytic green ammonia synthesis

Organic small molecule amidation
Doctoral candidates

Photon-phonon co-driven CO2 conversion

Photon-phonon co-driven catalytic NH3 synthesis

Photon-phonon co-driven methane conversion

Photon-phonon co-driven methanol conversion

Photon-phonon co-driven ammonia synthesis

Photon-phonon co-driven C-H activation

Ultrafast spectroscopic analysis of carrier dynamics in photon-phonon co-driven catalysis

Time-resolved molecular vibration spectroscopy

Photon-phonon co-driven catalytic C-F bond activation
Undergraduates

Photon-phonon co-driven ammonia synthesis from N2 and H2O
Alumni

Methane conversion, ammonia synthesis, and plastic conversion

Energy and environmental photocatalysis with particular emphasis on the photocatalytic conversion of methane

Light-/Thermal-driven small molecule conversion
Publications in the Past 10 Years
2026
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
2015
2014
All Publications
News
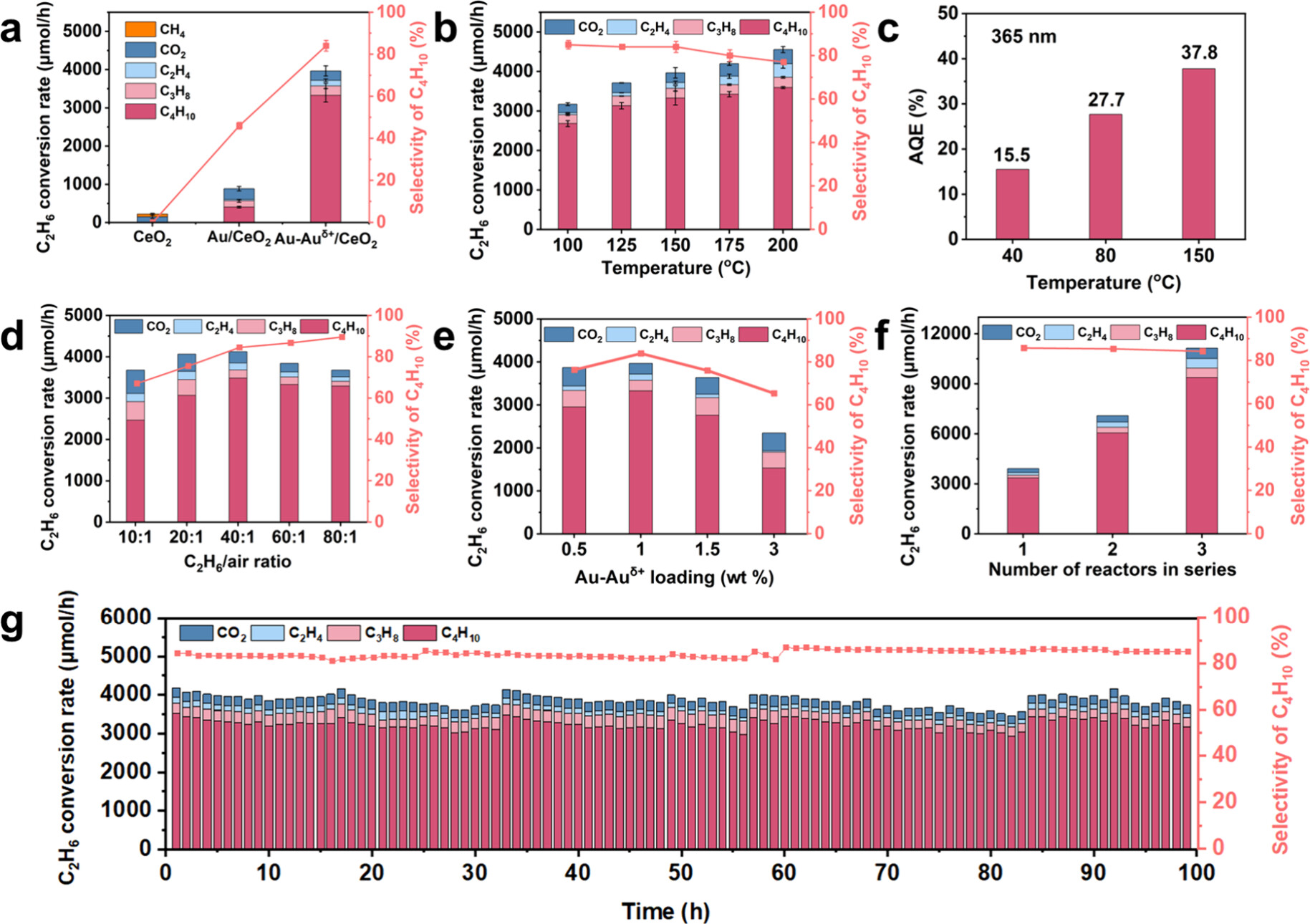
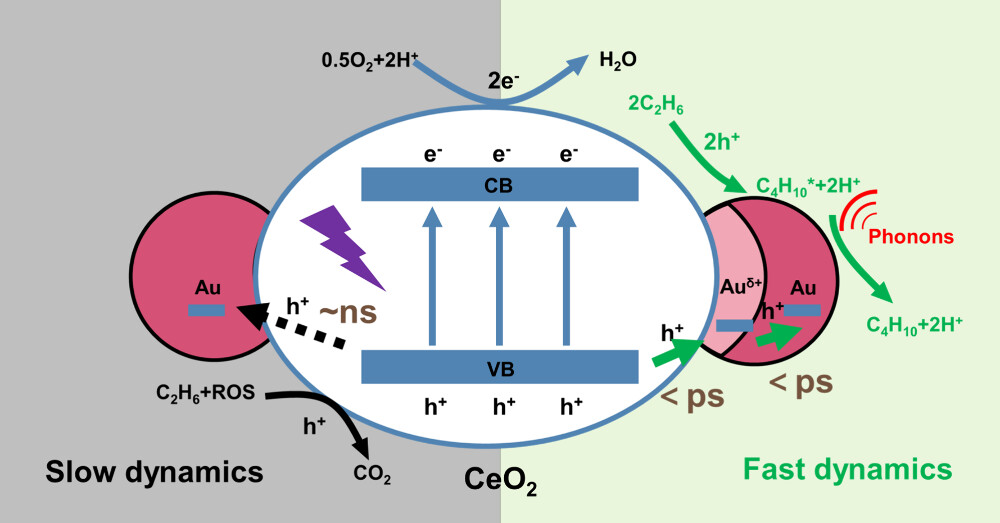
Our group published a research paper in Journal of the American Chemical Society: Auδ+–Au Relay-Promoted Synergy of Light Irradiation and Heating for Highly Selective Activation of C–H Bonds
A new study from the Tang group, demonstrates an elegant strategy to achieve highly selective C–H activation of ethane under mild conditions. By designing a Au–Auδ⁺/CeO₂ catalyst, first author Qin, Ziying and colleagues established a Auδ⁺–Au relay that accelerates hole transfer from the support to the active sites by 1000‑fold—from nanoseconds down to picoseconds. This ultrafast charge separation suppresses unwanted C–C cleavage and overoxidation, directing holes precisely toward the desired C–H activation.
Harnessing the synergy of light irradiation (photons) and heating (mainly phonons) , the system delivers a butane production rate of 166.5 mmol/g/h with 85% selectivity at a high space velocity, along with a turnover number of 410,000 and excellent stability over 100 hours. The process scales readily while maintaining performance and achieves an apparent quantum efficiency of 38%. This work provides a powerful example of how multifield coupling—photons, phonons, and charge relays—can be orchestrated to enable low‑carbon, sustainable synthesis of valuable chemicals.
Link: https://doi.org/10.1021/jacs.5c19973

Our Breakthrough in Photo-thermal Co-driven Catalytic Methanol-to-Hydrogen Featured in CCTV Special Report
Recently, our innovative achievement on photothermal synergistic catalytic methanol reforming for hydrogen production, was featured in a special report by CCTV News as a landmark achievement of the first batch of the "Non-Consensus Project" funded by the Beijing Municipal Natural Science Foundation.
The novel photothermal synergistic catalysis strategy proposed by the team achieves a hydrogen production rate from methanol reforming over 7 times higher than that of the state-of-the-art thermal catalysis technology under mild reaction conditions. Meanwhile, it successfully converts the oxidation product of methanol from carbon dioxide to high-value formaldehyde, realizing the co-production of clean hydrogen energy and high value-added chemicals, and effectively overcomes the core bottlenecks of traditional thermal catalytic hydrogen production processes, including high energy consumption, high carbon emissions, and poor catalyst stability. This achievement provides a new route for the development of green hydrogen production technology, and will offer critical technical support for the realization of China’s "Dual Carbon" goals.
Link: https://content-static.cctvnews.cctv.com/snow-book/video.html?item_id=2910678599157402924&t=1774074803167&toc_style_id=video_default&share_to=wechat&track_id=256dc780-b39d-4922-b6e0-b24f1aeefdb0



Our Breakthrough in Photo-thermal Co-driven Catalytic Methanol-to-Hydrogen Featured in Beijing Daily and Science and Technology Daily
We are pleased to share that our team’s work on innovative solar hydrogen production has been recently featured in two leading national publications: Beijing Daily and Science and Technology Daily.
These reports highlight our development of a novel photothermal catalytic hydrogen production pathway and acknowledge the forward-looking support from Beijing’s “Non-Consensus Innovation” funding program. Being featured in these influential media outlets reflects the broader interest and significance of our approach to sustainable energy. We are encouraged by this attention and remain committed to advancing clean hydrogen technology.
Links:
Beijing Daily: https://news.bjd.com.cn/2026/02/07/11570616.shtml
Science and Technology Daily: https://epaper.stdaily.com/statics/technology-site/index.html#/home?isDetail=1¤tNewsId=a9e8a67d39d0439790664f9c4e64b298¤tVersionName=%E7%AC%AC03%E7%89%88%EF%BC%9A%E8%A6%81+%E9%97%BB¤tVersion=3&timeValue=2026-02-04

Tang Group Wraps Up Annual Meeting with Focused Discussions
Our group successfully held its annual year-end meeting on January 24 at Room 204, Yingshi Building. The meeting was dedicated to reviewing the year's progress and planning future research directions.
Doctoral students and postdoctoral fellows presented their key research findings, challenges, and ongoing work. The session concluded with a presentation and guidance from Prof. Tang, Junwang, outlining priorities and objectives for the upcoming year.
Following the meeting, the group gathered for a lunch to celebrate a year of hard work and collaboration. We thank all members for their contributions and look forward to advancing our research in the coming months.



Dr. Yu, Yang and Dr. Chen, Yong from Sinopec Nanjing Institute and Tang Group Hold Project Exchange Meeting on Green Ammonia
On January 13, Sinopec Nanjing Chemical Industry Research Institute Co., Ltd. and Tsinghua University held a project exchange meeting at Tsinghua University. The meeting focused on the collaborative research progress for "Green Ammonia Synthesis under Mild Conditions."
Both parties reviewed the project's advancements, discussed key technical challenges such as catalyst optimization and industrialization pathways, and planned the next steps for research and development. The meeting concluded with a clear division of tasks to ensure the project's high-standard completion.
This regular exchange further solidifies the industry-academia partnership, driving the joint project forward toward its application goals.

Dr. Feng, Fan Selected for Shuimu Tsinghua Scholar Program
We are pleased to announce that Dr. Feng, Fan from our group has been selected for the second batch of 2025 Shuimu Tsinghua Scholar Program.
The Shuimu Tsinghua Scholar Program, established in 2019, is Tsinghua University's flagship initiative to attract and cultivate outstanding early-career researchers. It provides selected scholars with a high-level development platform, comprehensive training, and competitive support, including an annual stipend and research funding. This prestigious selection recognizes the researcher's academic excellence and research potential. It will provide valuable resources and opportunities to further advance their work within our group's research projects.
We extend our warmest congratulations and look forward to their continued contributions to our team's scientific endeavors.
Dr. Yang, Jianlong Successfully Defends Postdoctoral Research at Tsinghua University
On December 23, Dr. Yang, Jianlong successfully defended his postdoctoral research at Tsinghua University. The defense committee was chaired by Academician Guo, Lin and included Professors Zhao, Lidong, Li, Chun, and Tang, Junwang.
Dr. Yang presented his work on "Modulation of TiO₂ Interface and Crystal Facets for Photocatalytic Methane Conversion." His research focused on designing and optimizing TiO₂-based photocatalysts to enhance efficiency and selectivity in converting methane into valuable chemicals. The two main projects involved synergistic crystal facet engineering with Pd nanoclusters and constructing a chemical cycling system to avoid over-oxidation.
Following the presentation, the committee engaged in a thorough discussion, providing valuable insights and suggestions for further deepening the mechanistic understanding and experimental design. The committee acknowledged the significance of the research and approved the defense.
We congratulate Dr. Yang on this important milestone and wish him continued success in his future scientific career.






Our group published a research paper in Advanced Materials: Infrared and Thermo Co-Driven Catalysis for CO2 Conversion to Valuable Chemicals
Our group has achieved a breakthrough in converting CO2 into dimethyl carbonate (DMC). By utilizing a novel photon-thermal synergy process with a defect-engineered, noble-metal-free cerium oxide catalyst, first author Chen, Enqi and colleagues have attained a remarkable DMC production rate of 30 mmol/g/h with 100% selectivity. This work overcomes the performance bottlenecks of conventional thermal catalysis and introduces a new strategy of IR-driven catalysis for CO2 utilization.
Link: https://advanced.onlinelibrary.wiley.com/doi/10.1002/adma.202512626



The research group achieves fruitful results at the 18th National Solar Photochemistry and Photocatalysis Conference
Our group achieved fruitful results at the 18th National Conference on Solar Photochemistry and Photocatalysis held in Tianjin. Professor Tang, Junwang present a Plenary Lecture titled "From Photocatalysis to Photon-Phonon Co-Driven Catalysis," which garnered significant attention from attending experts. Dr. Xiong, Lunqiao presented an oral presentation in a parallel session entitled "Application of Single-Atom/Atom Cluster Photocatalysts in Glycerol Oxidation." Furthermore, the academic poster presented by Zheng, Yaxuan, received the Best Poster Award.
Link: https://news.tju.edu.cn/info/1012/566149.htm



Professor Junwang Tang was awarded the International Science Award for Crossing Boundaries by Falling Walls Foundation
Professor Junwang Tang has been named a 2025 Falling Walls Breakthrough Winner in Engineering and Technology. This prestigious award, presented by Germany's Falling Walls Foundation, honors top scientific breakthroughs selected from more than 1,500 international nominations.
Link: https://falling-walls.com/engineering-technology
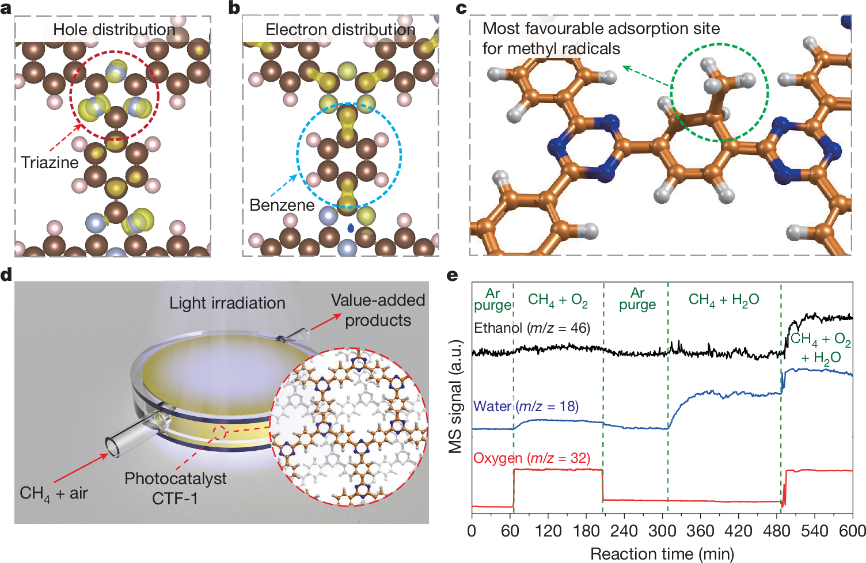
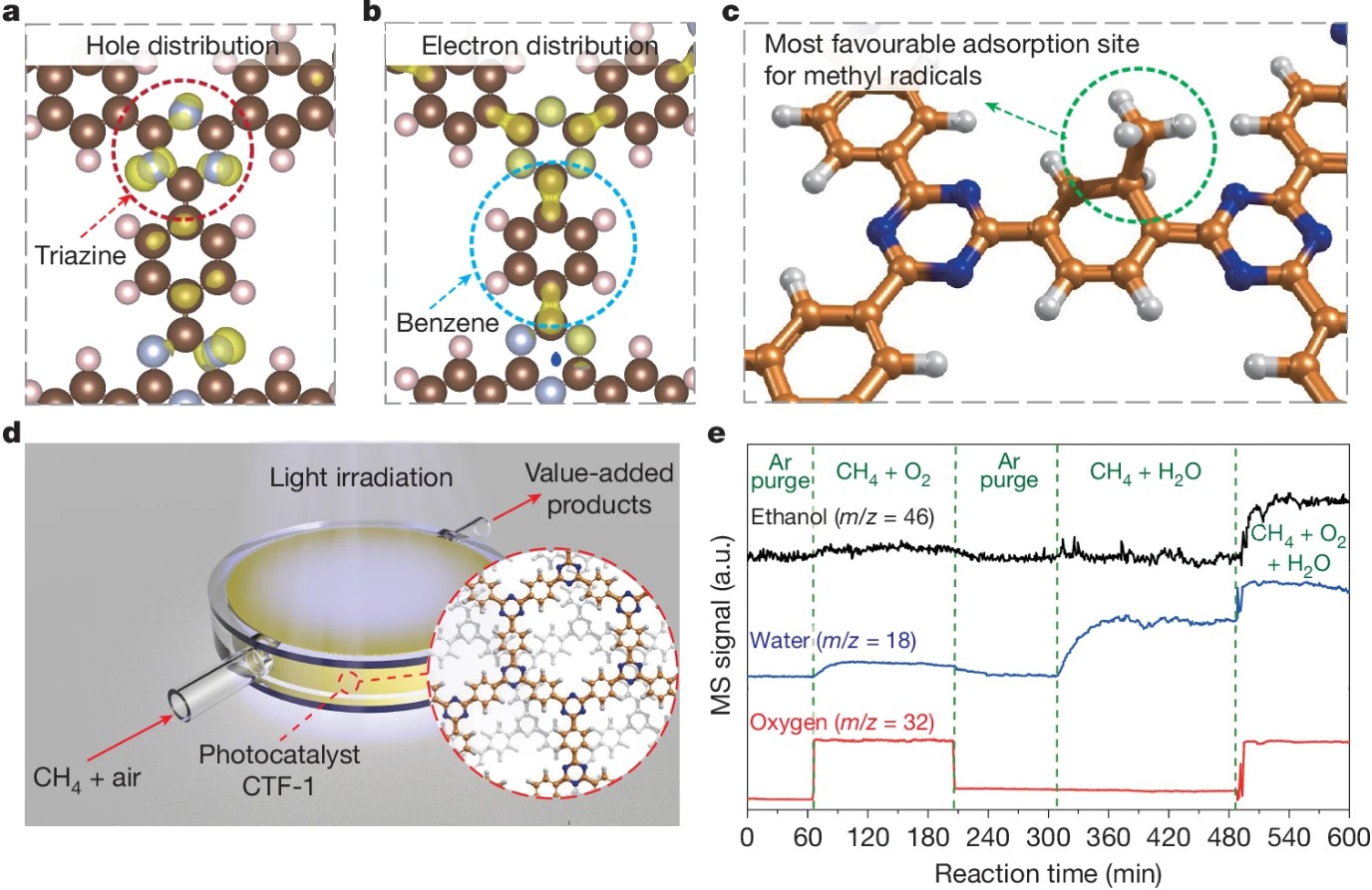
Our group published a research paper in Nature: Methane Oxidation to Ethanol by a Molecular Junction Photocatalyst
Professor Tang Junwang's team proposed a novel concept of molecular junction and developed the CTF-1 catalyst. Leveraging the synergy between triazine and benzene ring units, this catalyst enables the highly efficient, one-step conversion of methane to ethanol at room temperature and pressure under light irradiation. This technology provides a groundbreaking pathway for the high-value utilization of natural gas resources, which not only helps reduce the chemical industry's heavy reliance on petroleum but also contributes to achieving carbon neutrality goals.
This achievement is the result of nearly a decade of highly efficient collaboration among a multinational team comprising five universities from China and the UK, highlighting the essential role of global scientific cooperation in addressing critical energy challenges. The related research, entitled "Methane oxidation to ethanol by a molecular junction photocatalyst," was published online in Nature on January 20 (Nature 2025, 639, 368–374).
Link: https://www.nature.com/articles/s41586-025-08630-x

Equipment
Our laboratory is equipped with state-of-the-art facilities for thermal, photon-phonon co-driven and microwave-assisted catalysis research. Some are listed below.
Ammonia Synthesis/Decomposition

Ammonia synthesis reaction evaluation devices

Gas compression device

Photon-phonon co-driven ammonia synthesis reaction system

Thermal catalytic ammonia decomposition reaction system

Photon-phonon co-driven ammonia synthesis reaction system

Photon-phonon co-driven nitrogen oxidation reaction system

Photon-phonon co-driven ammonia synthesis reaction system

Photon-phonon co-driven ammonia synthesis reaction system

Photon-phonon co-driven ammonia synthesis reaction system with 50×sun light intensity Xe light source

Ultrasonic-photon coupled catalytic ammonia synthesis reaction system

Ion chromatography
Solar-driven Hydrogen Production

Photon-phonon co-driven methanol reforming reaction system

Photon-phonon co-driven methanol reforming reaction system

Gas chromatography
Photon-phonon Co-driven Biomass Conversion

Photon-phonon co-driven methanol conversion reaction system

Photon-phonon co-driven glycerol conversion device

Gas chromatography-mass spectrometry
Microwave-assisted Catalysis of Plastic Depolymerisation

Microwave assisted catalysis of plastic degradation reaction system
Photon-phonon Co-driven Natural Gas Conversion



3 setups for photo-phonon co-driven natural gas conversion

Phton-phonon co-driven methane combustion evaluation device

Gas chromatography

Gas chromatography
Multi-scale Time Resolved Spectroscopy


Time-resolved ultrafast spectroscopy platform for photophysics and reaction kinetics analysis [transient absorption spectrometer (TAS); time-resolved infrared spectrometer (TRIR); femtosecond stimulated Raman spectrometer (FSRS)]
Materials Synthesis and Characterisation

Uv-vis-nir absorption spectrometer

Photoluminescence spectrometer

Tube furnace

Air drying oven

Air drying oven

Vacuum oven

Vacuum oven

Muffle furnace

Centrifuge
Galleries












